Valence electron groups periodic table3/31/2024 
Unfortunately, there was a slightly different system in place in Europe. Atoms share electrons and form covalent bonds to satisfy the octet rule. 
4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. The atoms in group 6A make two covalent bonds. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. Covalent bonds are formed by two atoms sharing electrons. The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. The trend for electron affinity values across periods and down groups is shown in this version of the periodic table. With the exception of groups 312 (the transition metals ), the units digit of the group number identifies how many valence electrons are associated with a neutral atom of an element listed under. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. The s-, p-, and d-block elements of the periodic table are arranged into 18 numbered columns, or groups. The electron configurations of the elements are in Figure 6.9.2. Atoms in Groups 13 and 18 have 3 and 8 valence electrons. 1: The Periodic Table, Showing How the Elements Are Grouped According to the Kind of Subshell ( s, p, d, f) Being Filled with Electrons in the Valence Shell of Each Element. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. The periodic table shows the atomic mass and atomic number of each element. 1: (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. Take a closer look at the some of the elements in the periodic table’s far right column in Figure 3. 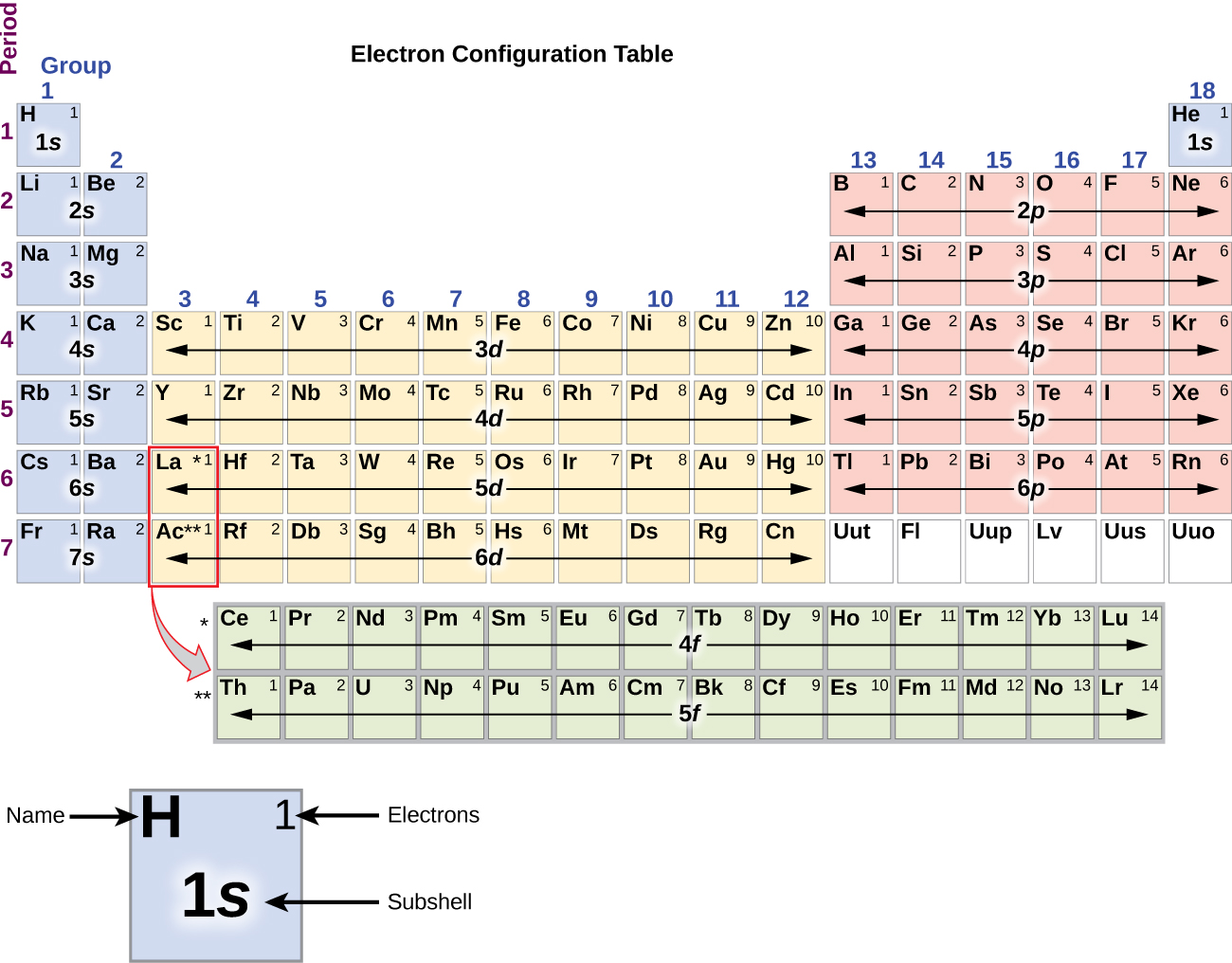
For example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively. The periodic table is arranged in columns and rows based on the number of electrons and where these electrons are located. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table. A new period begins when a new principal energy level begins filling with electrons. Valence electrons are the electrons present in the outermost shell of an atom. There are seven periods in the periodic table, with each one beginning at the far left. Elements whose atoms have the same number of valence electrons are grouped together in the Periodic Table. Solid nonmetals are also very brittle.\) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table. for p-block elements, the group number is 10 plus the number of valence electrons. For s-block elements, the group number is the number of valence electrons. Moving down a group adds a new electron subshell. Groups are the vertical columns on the periodic table. Each new period begins with one valence electron. The rule is as follows: If an element is not a transition metal, then valence electrons increase in number as you count groups left to right, along a period. A nonmetal is typically dull and a poor conductor of electricity and heat. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. Blocks indicate which electron sublevel is being filled. Apply the rule of the periodic table to your element. Metals are also malleable (they can be beaten into thin sheets) and ductile (they can be drawn into thin wires). A metal is a substance that is shiny, typically (but not always) silvery in color, and an excellent conductor of electricity and heat.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
